What is HypoxyLab™?
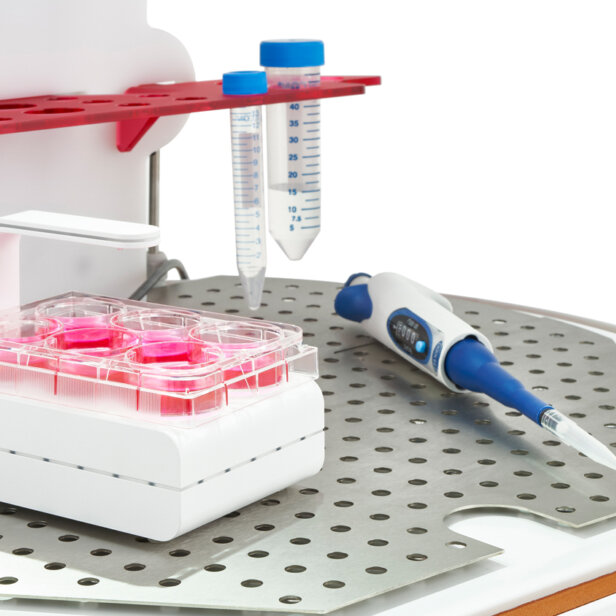
HypoxyLab is a cell culture incubator and hands-on workstation that accurately mimics and maintains true physiological oxygen conditions, as well as maintaining temperature, humidity and CO2 conditions like any standard cell culture incubator.
How does HypoxyLab™ differ from other hypoxia workstations?
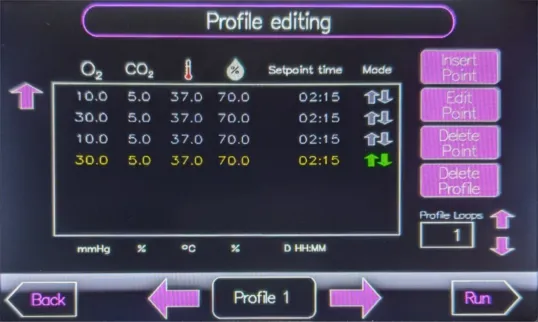
The HypoxyLab is sufficiently compact to fit on an ordinary laboratory bench, does not require the use of a dedicated transfer hatch, and comes fitted from the factory with built-in HEPA filtration, active humidification control, and an oxygen profiling feature. Additionally, the HypoxyLab regulates its oxygen environment using the partial pressure of oxygen (in mmHg) for the ultimate in scientifically rigorous oxygen control precision.
Why mmHg instead of % oxygen?
Using the partial pressure of oxygen to control the oxygen environment within HypoxyLab is simply the more scientifically rigorous thing to do! Since atmospheric (barometric) pressure changes with weather patterns, as well as with laboratory altitude, so does the oxygen concentration if it is merely maintained as a percentage thereof. Instead, HypoxyLab uses the partial pressure of oxygen to compensate for barometric pressure, ensuring that cells cultured within it 'experience' a consistent oxygen concentration whatever the weather and laboratory location. It ensures that data generated in HypoxyLab can be compared with and/or accurately reproduced by any other HypoxyLab user around the world.
How do I get items in and out of the system?
Cell culture plates, medium, pipettors and other objects can be moved in an out of the HypoxyLab via the simple to operate easy-entry hatch at the front, or can be placed in the chamber prior to the start of the experiment by simply lifting off the whole enclosure cover.
How does the entry hatch prevent ambient oxygen from entering the HypoxyLab™?
Opening of the hatch is accompanied by a transient internal overpressure generated by injecting nitrogen. This completely prevents ingress of external air in most scenarios. Only under extreme hypoxia, or where the hatch is opened for an unusually long time, will a small oxygen transient be observable. If so, this is eliminated within 2-3 minutes, ensuring no adverse effects on cell cultures.
Do I need to wear gloves to operate HypoxyLab?
There is no requirement per se for the operator to wear gloves, however, as with all cell culture work, wearing gloves represents good laboratory practice.
How can I clean the HypoxyLab™?
The HypoxyLab can be easily cleaned/disinfected using 70% ethanol or hydrogen peroxide based surface cleaners by removing the enclosure cover to expose all internal surfaces.
What gases are required to operate HypoxyLab™?
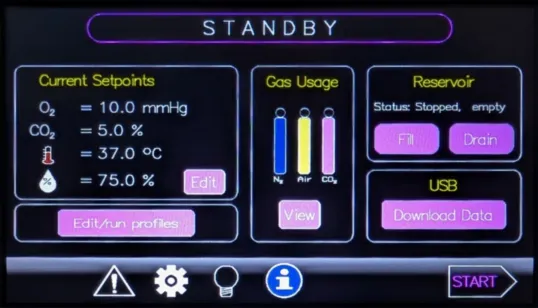
HypoxyLab requires a source of 3 gases, namely 100% nitrogen, 100% CO2, and synthetic air (typically 20% oxygen, balanced in nitrogen). If the oxygen profiling feature or rapid increases in internal oxygen are not a requirement, the HypoxyLab can optionally operate on just CO2 and nitrogen alone. All gases should be laboratory grade (99.98% / 'N3.8' or better) and must be supplied at an inlet pressure of between 1 - 5 bar (15 - 70 PSI).
Is the user protected from substances inside the HypoxyLab™?
No. While the HypoxyLab features a built-in HEPA filter that continuously ‘scrubs’ the internal atmosphere, the system does vent externally. The HypoxyLab is NOT therefore a substitute for a fume cupboard or a biological safety cabinet.
How clean is the internal environment?
The built-in HEPA filtration system achieves an atmosphere equivalent to ISO 14644-1 Class 2 within a few minutes of power-up, helping to protect cultures and media from the risk of contamination.
Can I measure dissolved oxygen levels directly from within my cell cultures?
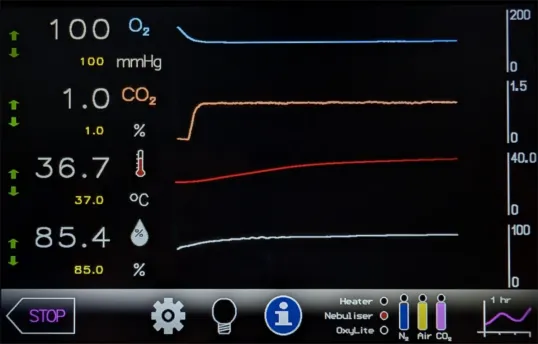
Yes. The HypoxyLab features a built-in gland that supports the introduction through the enclosure of our optional OxyLite™ fibre-optic dissolved oxygen sensors. These can provide a reading of dissolved oxygen in units of mmHg, directly from media or cell cultures within the chamber.
Does the user need to be standing to operate the HypoxyLab™?
The system can be operated standing up or from a seated position. A height-adjustable stool is recommended for seated operation.
What is the typical nitrogen consumption rate of the HypoxyLab™?
Nitrogen consumption is extremely dependent on the conditions being maintained and the usage ‘profile’ being employed. Typically, a standard (50L) laboratory gas cylinder of compressed nitrogen can be expected to support 2-6 weeks of continuous use.
Do I have access to the trace data displayed on the touchscreen?
Yes. Trace data are automatically and continuously stored to internal memory and can be copied to USB media at any time. Data are written in a format compatible with LabChart Reader software, a free viewer from ADInstruments.