Introduction
Exploring the domain of hypoxia or physioxia research demands a level of complexity beyond that which meets the eye. It demands significantly more thought than does the use of a standard incubator, where cells are simply maintained at 37°C, high humidity, and 5% CO2 for a set duration.
This article aims to shed light on critical considerations for researchers, especially those new to hypoxic or physioxic studies. By scrutinizing oxygen concentrations both inside and outside of biological incubators and delving into the significance of chamber atmosphere and pericellular oxygen levels, we aim to provide valuable insights for those new to these studies.
CO2 incubators deviate from 21% oxygen
To draw meaningful comparisons between controlled conditions in a hypoxia workstation and the conventional cell culture environment, it is vital to recognize that even a standard CO2 incubator does not maintain a 21% oxygen environment. Instead, the result of maintaining a high humidity and 5% CO2 environment is that the oxygen concentration inside an incubator is significantly altered.

Water vapor displaces other gases within the chamber, with a more pronounced effect at higher altitudes due to reduced gas mass. Studies have revealed that oxygen levels inside standard CO2 incubators hover at or around 17%. Moreover, opening and closing the incubator door can cause significant fluctuations in oxygen levels, often raising them to 21% within 20-30 seconds. Since standard incubators do not monitor or actively control oxygen levels, incubated cells are being exposed to oxygen levels that can potentially vary between 15-21%.
While hypoxia workstations are intended to mimic physiological oxygen concentrations (which are typically in the 1 - 5% oxygen range), any scientist using one to recreate the conditions of a standard incubator would we advised to aim for the 16-18% oxygen range, rather than the canonical 21% oxygen.
Are incubator oxygen levels suitable for physiological studies?
Physiological studies require a thorough understanding of the oxygen conditions that live cells encounter and it's important to note that very few examples exist of cells naturally exposed to oxygen levels above 17%. Such high oxygen exposure is primarily limited to superficial body surfaces, like the nasal epithelium or the oral cavity. Even in these locations cells are often dead, covered in mucus or fluids, and thus exposed to oxygen levels significantly lower than those in standard CO2 incubators.
When oxygen enters the body through the airways, it mixes rapidly with expired gases, reducing from 21% to approximately 10-13% by the time it reaches the alveoli. Different tissue types within the body operate within oxygen ranges that deviate significantly from room air conditions, as highlighted in the extensive 2018 review by Keeley and Mann, “Defining physiological normoxia for improved translation of cell physiology to animal models and humans", and summarized in the table below (taken from “Lost in application: Measuring hypoxia for radiotherapy optimisation”).
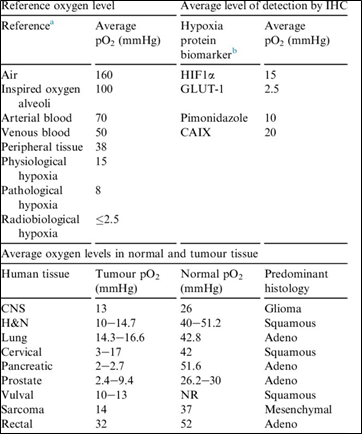
The review above not only explores many relevant in vivo oxygen ranges but also emphasizes the challenge of reproducing oxygen conditions when it is expressed in percent, as this does not account for the variables of laboratory altitude or day to day atmospheric (barometric) conditions - see our related article, “The reproducibility issue within hypoxia chambers and a simple solution to fix it”.
Chamber atmosphere is not the only factor
The oxygen concentration within even an oxygen-controlled atmosphere may not accurately reflect the pericellular oxygen levels that cells experience. As cells consume oxygen in the culture medium, the dissolved oxygen levels can drop significantly compared to the gas atmosphere in the vessel headspace. For instance, when the medium is equilibrated to an atmosphere of 1% O2, pericellular oxygen may rapidly decrease to near 0% O2 in the presence of cells.
The decline in pericellular oxygen is influenced by factors such as cell density, cell type, metabolic state, medium volume, agitation, and the type of cell culture vessel used. Consequently, the conditions experienced by cells can be highly protocol-dependent. The diffusion of chamber gases into the vessel headspace, subsequent dissolution in the medium, and final diffusion to the cells are slow processes due to the sluggish nature of oxygen dissolution in aqueous solutions.
To address this issue, Wenger et al.'s 2015 review offers a comprehensive examination of the numerous factors affecting pericellular oxygen levels. This paper challenges the widely accepted belief that room air incubators contain 21% oxygen and delves into the oxygen levels found in culture media, providing a deep understanding of the complexities involved in maintaining physiologically relevant conditions for cellular research.
Our recommendations for new users often encompass selecting appropriate plateware (with gas-permeable bottoms if possible) to enhance media exposure to the surrounding environment. Pre-conditioning media to match prior cell conditions is essential to avoid inducing cellular stress when exchanging media. Additionally, monitoring cell and media pO2 directly using tools such as the OxyLite monitor may prove helpful to figure out exactly what pO2 levels the cell layer is actually at.
Conclusions
In conclusion, delving into the intricacies of hypoxia and physioxia research demands a comprehensive understanding of the complexities surrounding oxygen concentrations. Our exploration of the challenges associated with CO2 incubator oxygen levels highlights the need for caution when comparing cell culture under controlled and uncontrolled oxygen conditions. Moreover, the examination of the relevance of oxygen levels in physiological studies emphasizes the importance of referencing comprehensive literature for appropriate oxygen ranges for the specific cells a researcher is using. Lastly, our insights into chamber atmosphere and pericellular oxygen levels shed light on the dynamic nature of oxygen in cell culture environments. To advance the field of cellular research while considering a molecule as important as oxygen, it is imperative for scientists to embrace a holistic approach, considering factors such as plateware selection, media preconditioning, and direct oxygen monitoring. By doing so, researchers can make informed decisions that enhance the accuracy and reproducibility of their experiments in the realms of hypoxia and physioxia.
System information
HypoxyLab – is a hypoxia/physoxia workstation used to mimic in vivo oxygen levels. The HypoxyLab allows manipulation within the system via sealed armports that ensure the maintenance of physiological oxygen conditions across an entire study. Users can control oxygen, carbon dioxide, humidity and temperature on a front facing touchscreen. Uniquely, the HypoxyLab ensures that oxygen levels are comparable and reproducible week to week and between laboratories in different geographical locations by maintaining and controlling internal oxygen conditions in absolute units of partial pressure (mmHg), as opposed to percent oxygen.

OxyLite – is our widely acclaimed tissue vitality monitor that measures pO2 in real time via an optical sensor. This is as simple as placing the sensor into the media (or against the cell layer) and waiting a few seconds for a reading to be made from the sensor tip.
Resources
Thomas P. Keeley and Giovanni E. Mann (2018).
Defining Physiological Normoxia for Improved Translation of Cell Physiology to Animal Models and Humans. Physiological Reviews
Thiruthaneeswaran N, Bibby B, .. and West C. (2021).
Lost in application: Measuring hypoxia for radiotherapy optimisation. Eur J Cancer
The reproducibility issue within hypoxia chambers and a simple solution to fix it (Oxford Optronix)
Wenger RH, Kurtcuoglu V, .. and Hoogewijs D (2015).
Frequently asked questions in hypoxia research. Hypoxia (Auckl)
Download this article
Author: Justin Croft, November 2023