Introduction
Dissolved oxygen (DO) measures the absolute amount of oxygen available in a fluid (such as blood plasma or interstitial fluid), often expressed as a partial pressure (e.g., mmHg) or concentration (e.g., µM).
In contrast, oxygen saturation (sO₂) represents the percentage to which haemoglobin binding sites are occupied by oxygen. In practical terms, DO reflects the actual oxygen tension in tissue, the oxygen unbound to haemoglobin and available for cells, while oxygen saturation indicates how fully loaded the carrier (haemoglobin) is with oxygen. DO thus offers direct measure of oxygen availability at the cellular level, making it more suitable for localized research applications where microenvironmental oxygen matters. Modern tissue oxygen monitors such as the OxyLite™ by Oxford Optronix provide continuous DO readings (tissue pO₂) in real time, enabling accurate, high-resolution oxygen sensing in vivo and in vitro.

Oxygen is fundamental to life, yet how we measure it depends greatly on context. In biomedical research – from tumour biology and neuroscience (stroke models) to tissue engineering, organ transplantation, and ischemia studies, the precision of oxygen measurement can directly impact data quality and interpretation.
Two commonly used terms, “dissolved oxygen” and “oxygen saturation,” are often assumed to be interchangeable. In reality, they describe very different aspects of oxygen status. Understanding this distinction is critical when selecting the right instrumentation and experimental approach for studies of tissue oxygenation or metabolism.
In this article, we clarify the differences between these two-oxygen metrics and explain why the OxyLite™ tissue oxygen monitoring platform is designed to measure dissolved oxygen (tissue pO₂) rather than oxygen saturation. This is not just a semantic distinction but a methodological one that has important consequences for accuracy and biological relevance when measuring tissue oxygen in research settings.
The relevance of oxygen in life science applications
Oxygen is a key variable in many physiological and pathological processes. In preclinical and translational research, accurate oxygen measurements provide deeper insight into cell metabolism, tissue viability, and disease progression, particularly in contexts where oxygen availability is limited or dynamically changing.
For example, in tumour biology, intratumoural hypoxia plays a central role in driving angiogenesis, therapeutic resistance, and metastatic potential. In neuroscience and stroke research, deficits in oxygen delivery and perfusion must be quantified precisely to model ischemic injury and evaluate neuroprotective strategies. Similarly, in tissue engineering and regenerative medicine, maintaining adequate oxygenation in 3D constructs is essential for cell survival and function. Even in areas like organ transplantation or trauma research, monitoring tissue oxygenation in organs can inform strategies to mitigate ischemia-reperfusion injury and improve graft survival.
Despite its importance, oxygen can be challenging to quantify reliably at the appropriate scale. The choice between measuring dissolved oxygen versus oxygen saturation has implications for sensitivity, spatial resolution, and biological interpretation. Selecting the correct metric depends on the research question and the environment of interest – whether it’s systemic circulation, isolated tissue, or a localized microenvironment such as the interstitial space within tissues.
Dissolved oxygen: an absolute metric of oxygen availability
Dissolved oxygen refers to the concentration or partial pressure of molecular oxygen (O₂) physically present in a fluid, such as interstitial fluid, plasma, or culture media, unbound to any carrier protein like haemoglobin. In physiological contexts, DO is often expressed as an oxygen tension in units of partial pressure (mmHg or kPa), which directly reflects the driving force for oxygen diffusion to cells. DO represents an absolute, quantitative measure of how much oxygen is freely available for metabolic use in the measured tissue or organ.
This metric is particularly relevant in cellular and microenvironmental contexts where oxygen diffusion and consumption create gradients. For example, in a 3D tissue scaffold or an in vivo tumour, DO levels can vary dramatically between well-perfused regions at the surface and poorly perfused, oxygen-deprived regions at the core. These spatial differences in oxygen tension drive local hypoxia, which in turn can alter cell signalling, gene expression, and responses to therapy. Unlike oxygen saturation (a relative metric tied to haemoglobin capacity), DO provides a direct snapshot of the actual oxygen pressure at the measurement site, independent of blood supply or haemoglobin concentration. This makes it invaluable in microenvironments where traditional systemic metrics fall short.
The OxyLite™ monitor uses a fibre-optic sensor based on fluorescence quenching to measure tissue DO (pO₂) in real time, with high spatial resolution and sensitivity. The miniature sensor is inserted into the tissue or sample, where it detects oxygen partial pressure without consuming oxygen itself. By tracking changes in the fluorescence lifetime at the probe tip, OxyLite™ outputs continuous readings of local pO₂. This allows researchers to monitor dynamic changes in oxygen availability directly within tissues or cultures. Although this approach is minimally invasive (requiring probe placement in the tissue), it yields direct, quantitative data on oxygen tension that non-invasive techniques cannot provide. The payoff is the ability to resolve fine-grained oxygen dynamics in situ. This data is essential for studying metabolism, hypoxia, or ischemia in living systems.
Oxygen saturation: A relative indicator of oxygen transport capacity
Oxygen saturation (sO₂) represents the proportion of available binding sites on haemoglobin (or another oxygen carrier, like myoglobin or synthetic carriers) that are occupied by oxygen. It is expressed as a percentage, reflecting how fully the carrier is loaded with O₂. In practice, “oxygen saturation” usually refers to haemoglobin saturation in blood. For example, arterial oxygen saturation (SaO₂ or SpO₂ as measured by pulse oximetry) indicates what percentage of haemoglobin in arterial blood is oxyhaemoglobin.
Oxygen saturation is widely used in clinical and physiological monitoring as a surrogate for systemic oxygenation. A pulse oximeter on a patient’s finger, for instance, noninvasively estimates arterial SpO₂ to help assess respiratory function and blood oxygen delivery in real time.
In those contexts, saturation is a useful indicator of how efficiently oxygen is being loaded onto haemoglobin in the lungs and transported to tissues. However, it’s important to remember that saturation is a relative measure – it depends on both the oxygen partial pressure and the haemoglobin’s binding capacity.
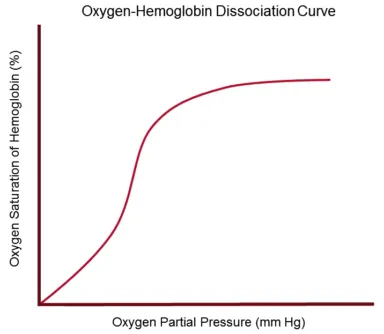
Several factors influence oxygen saturation readings. The haemoglobin–oxygen dissociation curve relates pO₂ to saturation in a sigmoid fashion, and its position shifts with pH, temperature, CO₂ levels, and other factors (the Bohr effect).
Moreover, two blood samples can exhibit the same % saturation but contain different absolute amounts of oxygen, if one individual has a higher haemoglobin concentration than another, or if one sample is at a higher pO₂ beyond the point of haemoglobin full saturation. In other words, 100% saturation does not equate to “100% oxygen” in a literal sense – it just means all available hemoglobin sites are occupied.
Because oxygen saturation hinges on carrier-bound oxygen, it is poorly suited for measuring oxygen at a local tissue level in many research scenarios. In avascular or low-perfusion environments – e.g. the centre of a tissue-engineered construct, the core of a tumour, or an isolated organ perfusion system – there may be little or no blood. A saturation value in such a context is either impossible to obtain or biologically meaningless, since it would read 0% in an anoxic region (no haemoglobin present), yet that doesn’t quantify how much O₂ is present for cells. Even within vascularized tissue, two regions could have identical blood oxygen saturation but different interstitial oxygen tensions, due to differences in perfusion or cellular uptake.
Dissolved oxygen vs oxygen saturation
In summary, while both “dissolved oxygen” and “oxygen saturation” describe aspects of oxygen status, they are fundamentally different in concept, measurement, and application:
- What is measured
Dissolved O₂ is an absolute measure – typically the partial pressure or concentration of free O₂ molecules at a point in fluid. Oxygen saturation is a relative measure – the percentage of oxygen-binding sites (on haemoglobin) that are occupied. One is a quantitative amount of oxygen; the other is a percentage of capacity used. - Units
DO is expressed in absolute units (mmHg, kPa, µM, mg/L, etc.), with mmHg (tissue oxygen tension) being common for physiological measurements. Saturation is expressed in relative units of percent (%) and inherently depends on haemoglobin concentration and binding dynamics. - Biological relevance
DO directly reflects the local oxygen environment – the O₂ available to cells for respiration at that location. It captures critical spatial gradients and diffusion limitations in tissues or cultures. In contrast, saturation reflects systemic oxygen transport. It’s most relevant in arterial blood or well-perfused tissues, indicating how fully blood is oxygenated overall. Saturation says little about actual availability of molecular oxygen to cells and tissue. - Measurement techniques
DO is measured with oxygen sensors that physically sample the tissue or medium. Oxygen saturation is typically measured with spectrophotometric methods – e.g. pulse oximeters or blood gas analyzers – that infer saturation from light absorption characteristics of oxy- vs. deoxyhaemoglobin. These require access to blood flow or extracted blood samples and cannot resolve spatial variability within tissue. - Spatial resolution
DO measurements can be made on the scale of microliters of tissue or even individual spheroids in vivo, potentially providing high spatial resolution. Oxygen saturation is usually a more coarse or global measure. - Data interpretation
Interpreting DO vs. oxygen saturation leads to different insights. It is possible for two tissue regions to exhibit the same haemoglobin oxygen saturation yet have very different dissolved oxygen levels. Relying on saturation to provide a measure of ‘tissue oxygen’ is misleading. DO provides a more direct and granular view of oxygen availability, especially in heterogeneous or ischemic tissues. Saturation, on the other hand, is excellent for monitoring respiratory function and overall oxygen delivery in an intact organism but might mask local hypoxia at the cellular level.
In research environments where oxygen availability at the tissue level is critical (hypoxia studies, ischemia models, etc.), measuring dissolved oxygen offers greater specificity and accuracy. Oxygen saturation remains valuable for clinical monitoring of patients and certain whole-body physiological studies, but it is often insufficient for microenvironmental research.
The OxyLite™ monitor measures dissolved oxygen
The OxyLite™ system was developed to meet a specific scientific need: quantifying oxygen at the tissue level in situations where traditional metrics like oxygen saturation are not scientifically useful.
Rather than measuring how much oxygen is bound to haemoglobin, OxyLite directly measures the oxygen tension (pO₂) in tissues, in units of mmHg. This approach provides a real-time readout of the oxygen available for cellular respiration in that region, essentially the balance between oxygen supply and consumption at the cellular scale.

This distinction matters in a wide range of research contexts. In tumour physiology, for instance, oxygen distribution is highly heterogeneous; local pockets of hypoxia can exist a few cell-layers away from well-oxygenated blood vessels. A saturation-based measurement would average out or miss these pockets, whereas the OxyLite probe can pinpoint them by measuring actual pO₂ in situ. In models of stroke or cardiac ischemia, tissue pO₂ can drop precipitously in the affected region even if blood SaO₂ is normal – the OxyLite can capture the true severity and timing of oxygen deprivation in the tissue. In 3D cell culture systems (spheroids, organoids, bioreactors), OxyLite sensors are often the only practical option, since there is no circulating blood to provide a saturation measurement. OxyLite sensors can also be inserted into culture vessels or even inside large spheroid/organoid models to monitor oxygen tension dynamically in the “microenvironment” that the cells actually experience.
Another advantage of DO measurements with OxyLite™ is its high temporal resolution and continuous monitoring capability. The system can detect rapid fluctuations in tissue oxygen levels (on the order of seconds) that would be averaged out with intermittent blood gas measurements.
The OxyLite platform has proven its value through extensive use by researchers worldwide. The OxyLite systems have been featured in hundreds of peer-reviewed publications, spanning fields from cancer biology to neuroscience and organ transplantation. Each of these studies capitalized on the ability to obtain quantitative, localized, physiologically relevant oxygen data that saturation-based tools couldn’t provide. By focusing on dissolved oxygen, OxyLite™ yields data that are more directly interpretable in terms of cellular biology.
It’s worth noting that measuring tissue DO with a microprobe is an invasive procedure, but only minimally so: the fibre-optic probe is fine and causes negligible tissue disruption. This is a reasonable trade-off given the wealth of information gained. In fact, for many applications, researchers consider it minimally invasive compared to alternatives like needle electrodes. The probe placement is typically done in anesthetized models or in excised tissue models.
Unlike electrode techniques of the past, the optical method employed by OxyLite™ does not consume oxygen during measurement, which ensures that the act of measuring doesn’t alter the local oxygen level.
Conclusions
Dissolved oxygen and oxygen saturation are both important measures in the study of oxygen biology, but they refer to fundamentally different concepts. Dissolved oxygen (DO), often expressed as a partial pressure (mmHg), quantifies the oxygen freely available in a liquid phase – it is an absolute measure of oxygen tension that directly reflects what cells have available to consume. Oxygen saturation, expressed as a percentage, indicates how much of a carrier’s oxygen capacity is utilized – it is a relative measure tied to haemoglobin binding and is most meaningful in the context of blood circulation.
Each metric has its realm of applicability. DO is the more appropriate metric for localized tissue oxygenation, hypoxic microenvironments, and in vitro systems where oxygen gradients and diffusion limitations are critical. It provides the specificity and resolution needed to map oxygen where it matters at the cellular level. Oxygen saturation is extremely useful for assessing systemic oxygen delivery in clinical or whole-animal scenarios, serving as a quick indicator of respiratory function and blood oxygen carrying status but it cannot reveal the fine detail of where oxygen is or isn’t in complex tissues.
For researchers in fields like oncology, neuroscience, physiology, or regenerative medicine, choosing the right oxygen metric is essential for obtaining meaningful data. The OxyLite system exemplifies a tool optimized for dissolved oxygen measurement, giving investigators a window into tissue-level oxygen dynamics that saturation-based tools cannot offer. By using OxyLite to monitor tissue pO₂, scientists can gain high-resolution insights into phenomena such as tumour hypoxia, ischemia, or organ perfusion, leading to more accurate models and potentially new therapeutic strategies. In short, when accuracy and biological relevance are paramount, measuring the oxygen that is available to cells (tissue oxygen) is often preferable to measuring how saturated the blood is.
When it comes to oxygen monitoring, one size does not fit all – so match your metric to your question; should you have questions about how the OxyLite™ might support your specific research needs, please contact us at sales@oxford-optronix.com. We’re here to help advance your oxygen-related research with the right tools and expertise.
Oxford Optronix is a supplier of world-class instruments for the life-scientist. Contact us to learn how our solutions can elevate your research.
Author: Justin Croft, 16 March 2026